ZC–modified sensor chips (Zwitterionic)
XanTec’s ZC sensor chips are coated with a zwitterionic polymer comprising an approximately equimolar ratio of carboxylic acid and tertiary amine groups. The resulting near-neutral net charge renders the surface highly bioinert toward charge-driven nonspecific binding.
ZC sensor chips support efficient EDC/NHS-based covalent coupling through a transient charge-shift mechanism. During activation, negatively charged carboxyl groups are converted into charge-neutral NHS esters, shifting the net surface charge toward positive. In this activated state, reversed-charge electrostatic preconcentration occurs, favoring enrichment of negatively charged ligands. After coupling, hydrolysis or quenching (e.g., with glycine) restores the charge-neutral zwitterionic surface.
These properties make ZC sensor chips particularly suitable for immobilizing acidic ligands, ligands unstable under acidic preconcentration conditions, and for analyzing strongly cationic analytes that would otherwise bind nonspecifically to classic polycarboxylate coatings such as CMD or HC.
Key features:
- Acidic ligand coupling: Reversed-charge electrostatic preconcentration enables covalent amine coupling of acidic ligands (e.g., DNA, RNA) at near-neutral pH.
- Gentle immobilization conditions: Efficient coupling typically around pH 6.5, preserving activity of pH-sensitive biomolecules.
- Wide immobilization range: Several thousand (ZC30M) to ~30,000 µRIU (ZC150D).
- Exceptional bioinertness: Charge-neutral zwitterionic matrix minimizes nonspecific binding, even for highly cationic biomolecules.
- No polysaccharide backbone: Absence of carbohydrate motifs prevents unintended lectin interactions.
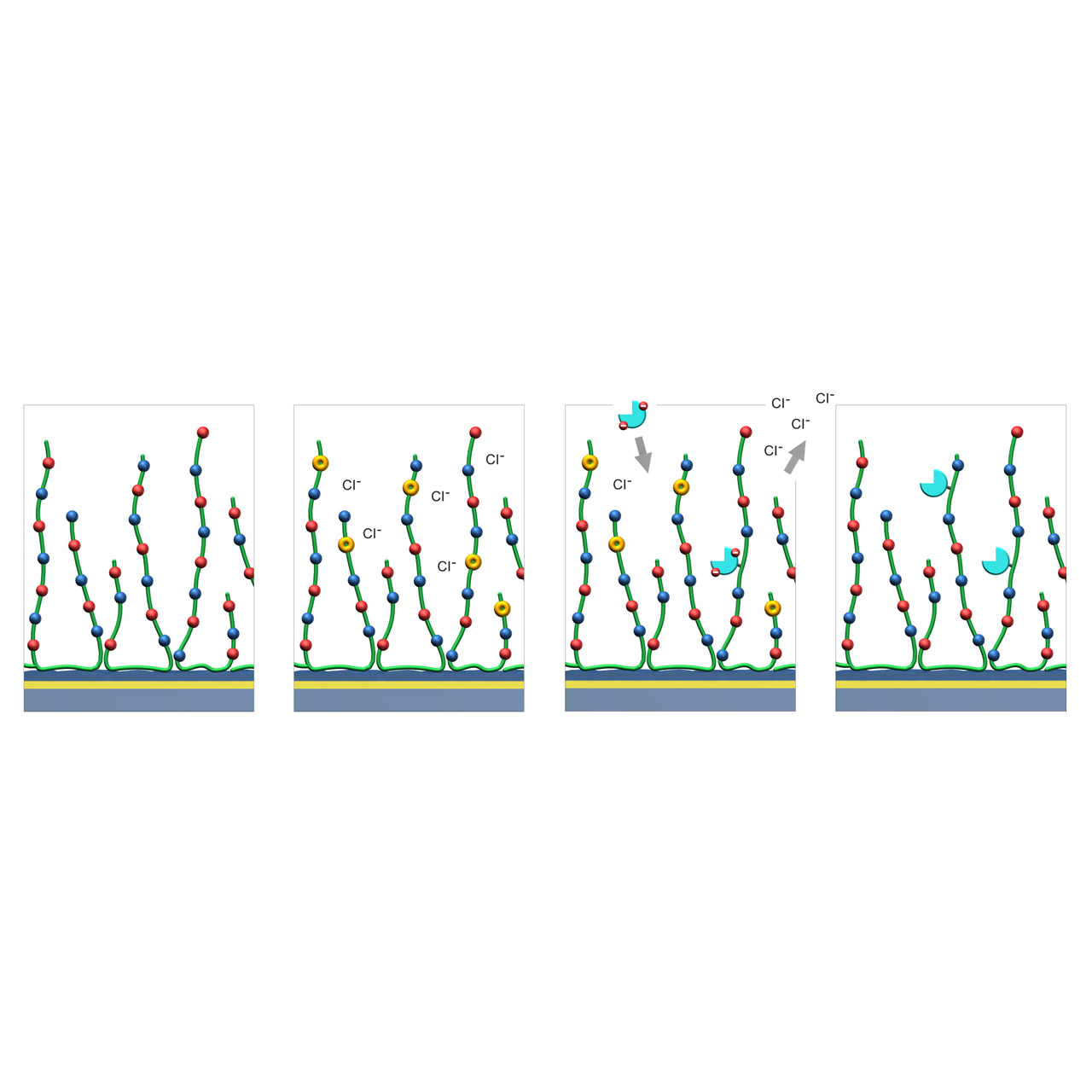
1. Native ZC sensor chip: Red and blue dots represent carboxyl and tertiary amine groups evenly distributed along the green polymer chains.
2. EDC/NHS activation: A fraction of the negatively charged carboxyl groups are converted into charge-neutral NHS esters (yellow), transiently shifting the net surface charge toward positive.
3. Reversed-charge preconcentration and coupling: Ligands with a negative net charge (cyan) accumulate at the surface via reversed-charge electrostatic preconcentration and are subsequently covalently immobilized through formation of amide bonds.
4. Deactivation: Hydrolysis or quenching (e.g., with 1 M glycine at pH 8.5) converts remaining NHS esters back into carboxylic acids, restoring the charge-neutral surface.
| Product code | ZC30M | ZC80M | ZC150D |
|---|---|---|---|
| Base coating | 3D, 30 nm bioinert charge-neutral zwitterionic polymer (medium density) | 3D, 80 nm bioinert charge-neutral zwitterionic polymer (medium density) | 3D, 150 nm bioinert charge-neutral zwitterionic polymer (high density) |
| Covalent immobilization capacity [µRIU]2 | ≈ 2,600 (3,000) | ≈ 6,000 (7,000) | ≈ 22,000 (30,000) |
| Recommended ligands3 |
|
||
| Recommended analytes |
|
|
|
| Intended purpose |
|
|
|
1 All illustrations are schematic representations and are not drawn to scale; dimensions, densities, and spatial relationships do not reflect actual physical or chemical proportions.
2 Typical covalent immobilization capacity and electrostatic preconcentration capacity (in brackets) determined by injecting 100 µg/mL Protein A in 10 mM MES buffer (pH 6.5) following surface activation with 0.5 M EDC in NHS/MES activation buffer.
3 All ligands require at least one NHS-reactive amino group.