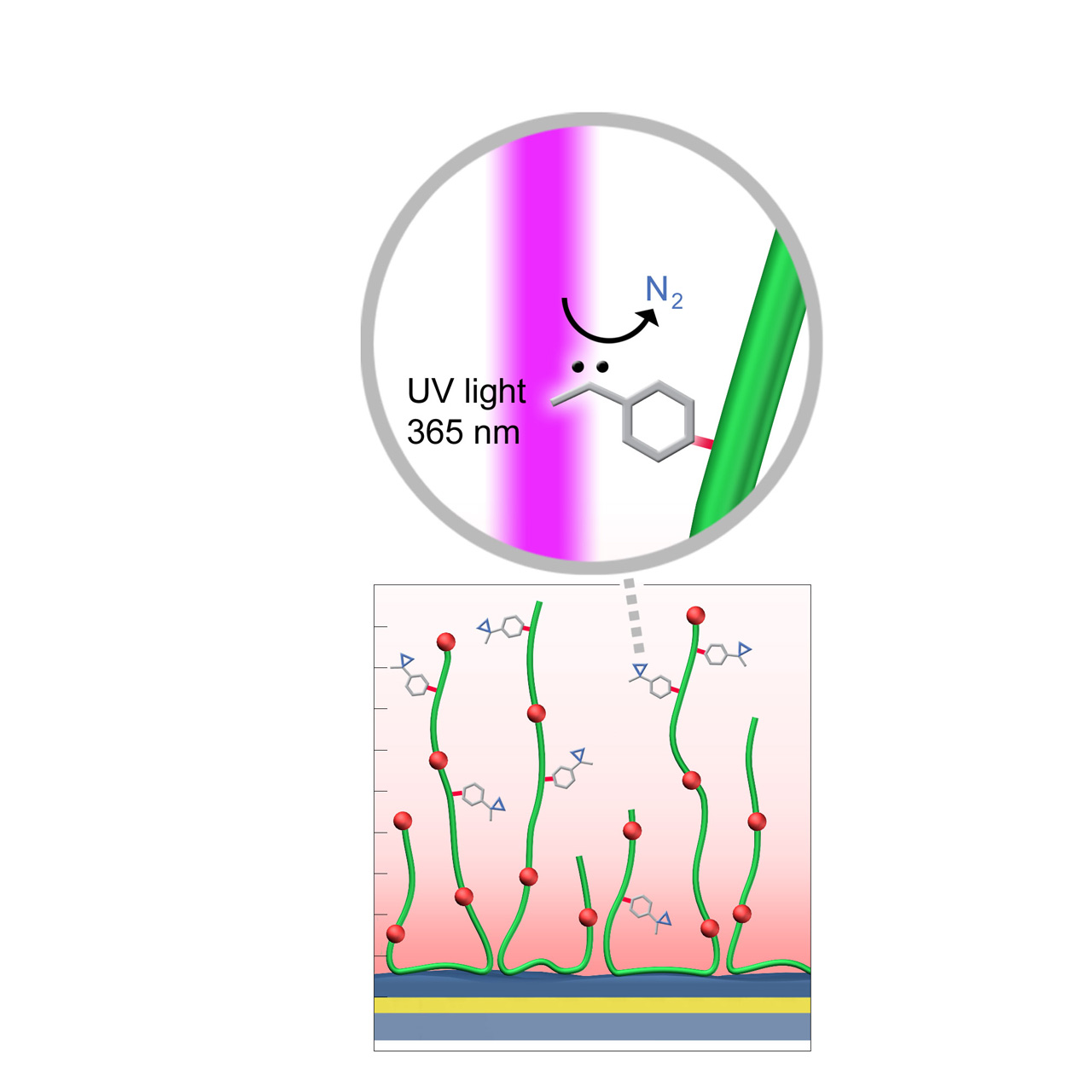
UV (Diazirine)–modified sensor chips
XanTec’s UV sensor chips are available as a 2D coating (UVHCP) or as 3D hydrogel matrices (UVHC200M, UVD200M) pre-functionalized with low-molecular-weight diazirine groups grafted onto a hydrophilic adhesion promoter on a gold support. Diazirines are stable in the dark but, upon irradiation at 365 nm, undergo photolysis, releasing N2 and generating a highly reactive carbene. This carbene inserts into thiol, amino, hydroxy, and even C–H bonds, enabling stable covalent attachment to virtually any biomolecule.
When combined with electrostatic preconcentration, UV coupling provides a robust and efficient immobilization strategy. Efficient immobilization requires direct UV light access to the sensor surface, which may necessitate a quartz flow cell window. In combination with microarray spotters, UV-activated coupling can also be used to generate highly defined microarray sensor chips or slides.
Key features:
- Robust covalent ligand coupling: Low chemical selectivity allows covalent attachment of nearly any biomolecule without additional reagents.
- Compatible with spot immobilization: Particularly suitable for microarray spotting and imaging-SPR applications; high coupling yields achievable after drying prior to UV activation.
- Requires special equipment: UV-permeable (quartz) flow cells are required for in-flow activation.
| Product code | UVHCP | UVHC200M | UVD200M |
|---|---|---|---|
| Base coating | 2D, ultra-short bioinert polycarboxylate (high density) |
3D, 1000 nm bioinert polycarboxylate (medium density) |
3D, 200 nm bioinert CM-dextran (medium density) |
| Covalent immobilization capacity [µRIU]2 | ≈ 1,500 | ≈ 11,000 | ≈ 10,000 |
| Recommended ligands |
|
|
|
| Recommended analytes |
|
|
|
| Intended purpose |
|
|
|
1 All illustrations are schematic representations and are not drawn to scale; dimensions, densities, and spatial relationships do not reflect actual physical or chemical proportions.
2 Covalent immobilization capacity determined by injecting 100 µg/mL BSA in 5 mM sodium acetate buffer (pH 5.0) and irradiating for 10 min with a 3 W, 365 nm UV LED using a quartz window flow cell.