Small Molecule Assay: 4-Carboxybenzenesulfonamide (201 Da) Binding to Carbonic Anhydrase II
Carbonic anhydrase II (CAII) is an enzyme that catalyzes the reversible hydration of carbon dioxide to form bicarbonate with the release of a proton. CAII has important physiological functions such as pH regulation, bicarbonate metabolism and control of intracellular osmotic pressure.1 CAII activity is strongly inhibited by a variety of aromatic and heterocyclic sulfonamides. This Application note presents the SPR binding experiment between CAII and an inhibitor, 4-carboxybenzenesulfonamide (4-CBS); a small molecule with a molecular weight of 201 Da.
Experimental
The experimental conditions for this assay are summarized below:
- Ligand: CAII
- Analyte: 4-CBS
- Analyte Concentrations: 20,6.7,2.2,0.74,0.25,0.082 μM
- Association Time: 1 min
- Dissociation Time: 3 min
- Regeneration Solution: none
Results
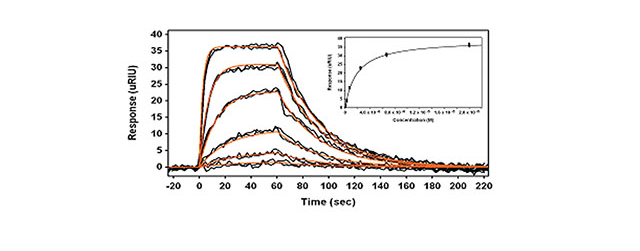
Figure 1 presents the kinetic results from this small molecule binding experiment. The inset represents the Langmuir binding isotherm where the equilibrium binding response is plotted as a function of concentration. Each concentration is injected at least twice to verify reproducibility. The kinetic data is fit to a simple bimolecular model using Scrubber (Biologic Software) (red lines) and the equilibrium data in the inset of Figure 1 is fit to a Langmuir binding isotherm model (solid line). This small molecule binding experiment was carried out on four separate occasions and Table 1 summarizes the results.
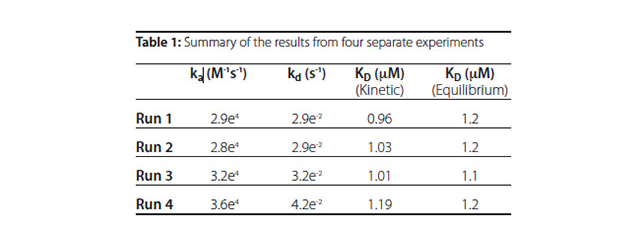
Table 1 presents the association and dissociation rate binding isotherm. constants determined from fits to a simple bimolecular model in Scrubber along with the equilibrium dissociation constants (KD) calculated from the kinetic and equilibrium data, respectively. The results show that the system is highly reproducible and the KD values determined through the kinetic and equilibrium analysis are in very good agreement with each other and correlate very well with that reported in the literature.