Strep-Tactin XT–modified sensor chips
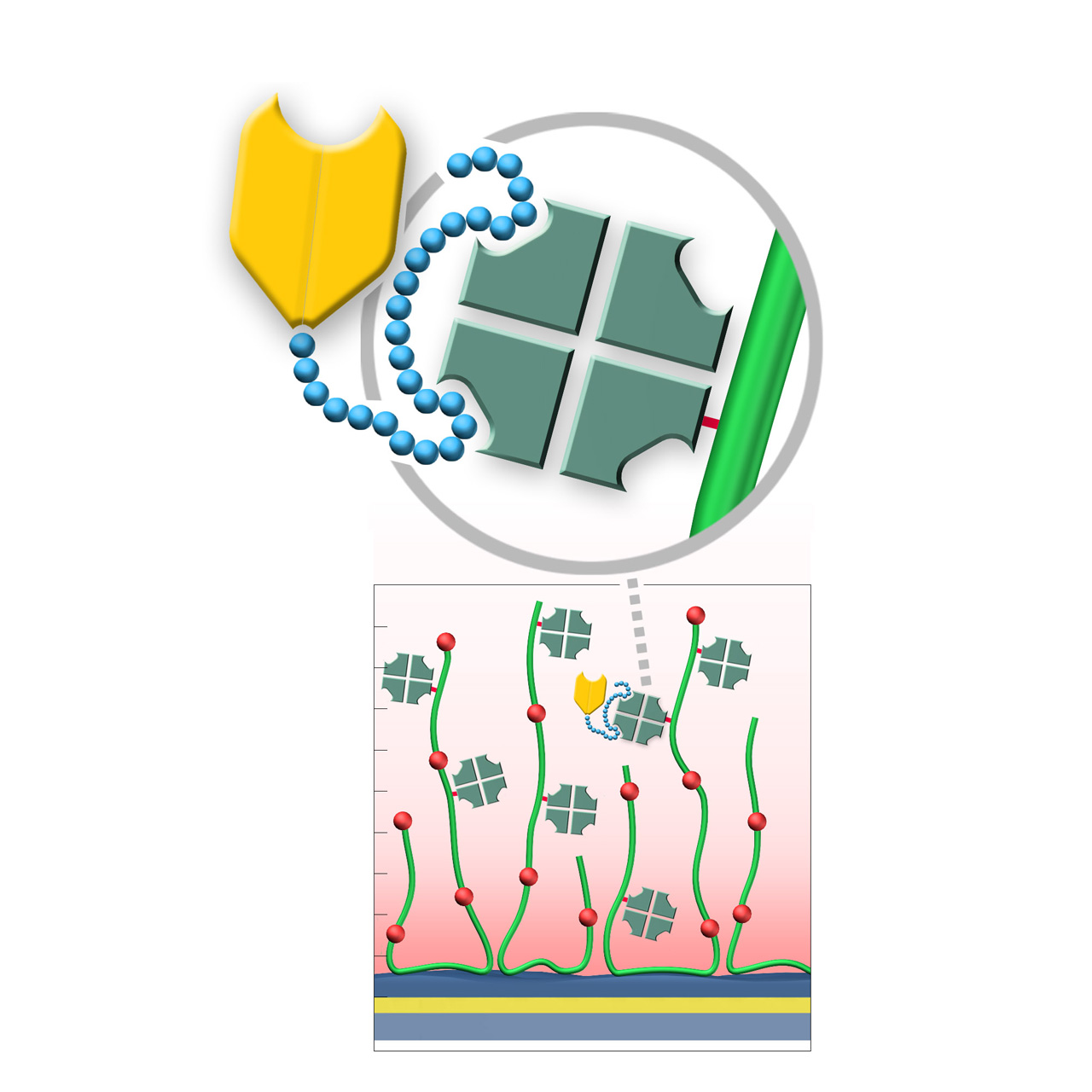
XanTec's Strep-Tactin XT-modified (ST) sensor chips are coated with a bioinert (poly)carboxylate matrix, pre-functionalized with a recombinant 52 kDa Strep-Tactin XT tetramer. Although a member of the avidin family, Strep-Tactin XT exhibits only weak micromolar affinity for biotin, making it unsuitable for stable immobilization of biotinylated biomolecules. However, it possesses exceptionally high picomolar affinity for the small 3 kDa Twin-Strep-Tag (TST) sequence, with typical dissociation rates (koff) of ≤ 10⁻⁵ s⁻¹.1,2 This allows defined, site-directed immobilization of TST fusion proteins under physiological conditions, ensuring uniform ligand orientation and preserving activity.
The Strep-Tactin XT/TST complex can be quantitatively regenerated using brief pulses of 3 M Guanidine·HCl, with ST chips tolerating more than 100 regeneration cycles. Compared with the NTA/His-tag system, ST chips display markedly lower nonspecific binding of proteins and peptides, making them ideal for kinetic and affinity studies involving these analytes. Together, these features establish ST sensor chips as an efficient and reliable tool for studying biomolecular interactions with TST fusion proteins, streamlining workflows and accelerating research.
XanTec offers three versions of Strep-Tactin XT–modified chips for SPR applications. The 2D STP chip provides superior diffusion properties, ideal for bulky analytes or weak binders with rapid kinetics, whereas the ST200L and STHC200M chips enable higher immobilization densities, well suited for smaller analytes.
Key features:
- Fast assay development: No preconcentration or chemical activation required. Controlled ligand capture occurs under physiological conditions, reducing preparation time and improving reproducibility.
- Exceptional stability: The strong picomolar Strep-Tactin XT–TST interaction ensures stable, virtually drift-free immobilization.
- Simple regeneration: Quantitative, rapid regeneration allows > 100 capture/regeneration cycles.
- Oriented immobilization: Uniform ligand orientation maximizes biological activity.
- Versatile capture capacity: Available on CMDP, CMD200L, and HC200M base coatings to match different assay requirements.
| Product code | STP | STD200L | STHC200M |
|---|---|---|---|
| Base coating | 2D, ultra-short CM-dextran (high density) |
3D, 200 nm bioinert CM-dextran (low density) |
3D, 200 nm bioinert polycarboxylate (medium density) |
| Capture immobilization capacity [µRIU]4 | ≈ 500–1,000 | ≈ 5,000–7,000 | ≈ 5,000–7,000 |
| Recommended ligands | TST-modified proteins and peptides | TST-modified proteins and peptides | TST-modified proteins and peptides |
| Recommended analytes |
|
|
|
| Intended purpose |
|
|
|
1 Apparent dissociation rate constants (koff) were determined at ligand capture densities < 50 % of the maximum immobilization level.
2 Binding of Strep-Tag II is feasible but the complex exhibits limited stability, with dissociation rates of koff ≈ 10⁻²–10⁻³ s⁻¹.
3 All illustrations are schematic representations and are not drawn to scale; dimensions, densities, and spatial relationships do not reflect actual physical or chemical proportions.
4 Based on the specific capture immobilization of 500 nM TST-GFP in PBS.