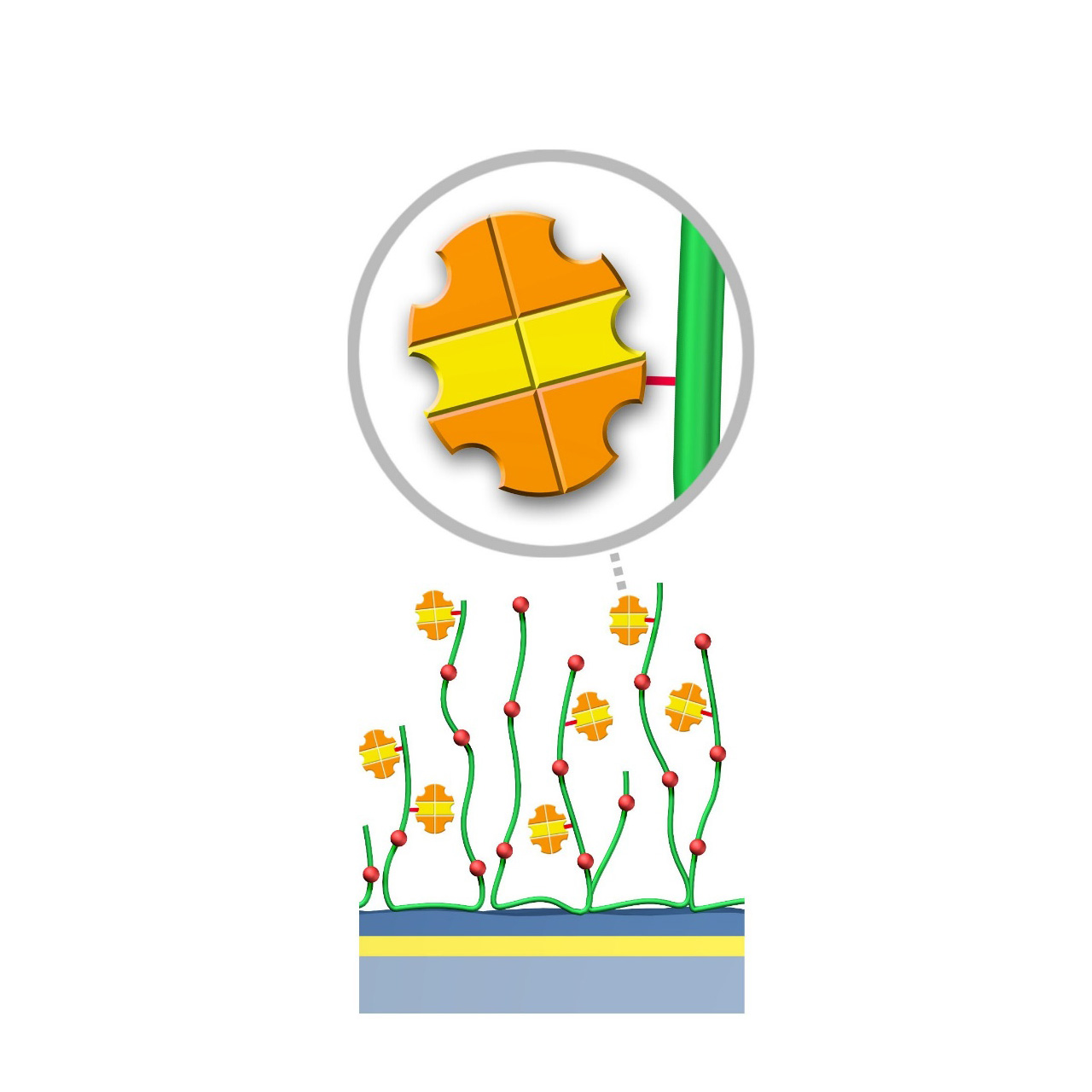
Protein AG–modified sensor chips
XanTec's PAGP, PAGD, and PAGHC sensor chips are coated with a bioinert polycarboxylate matrix pre-functionalized with recombinant 50.5 kDa Protein AG. This non-glycosylated Protein AG variant includes four IgG-binding domains derived of Protein A and two IgG-binding domains from Protein G, providing high-affinity binding to the Fc region of all human IgG subclasses. Additionally, it exhibits strong affinity for mouse IgG2a, IgG2b, and IgG3, as well as total IgG from other mammals including cow, goat, sheep, and rabbit.
To ensure highly specific IgG binding, non-essential domains—such as cell wall-binding, cell membrane-binding, and albumin-binding regions—have been removed from the recombinant Protein AG. Nevertheless, due to its slightly hydrophobic character, some nonspecific binding may still occur. The inclusion of blocking agents (e.g., BSA) and/or nonionic detergents (e.g., Tween) in the running buffer is therefore recommended to minimize background signals.
XanTec’s Protein AG sensor chips are ready-to-use, eliminating the need for time-intensive assay optimization and streamlining workflows. With five distinct Protein AG-modified versions available for SPR applications, each is tailored to specific analytical needs.
Key features:
- Fast assay development: No preconcentration or surface activation required. Controlled and reversible ligand immobilization is performed under physiological conditions, reducing preparation time and ensuring reproducibility.
- Broad antibody compatibility: Broader antibody subclass and species affinity than Protein A.
- Simple regeneration: Chip surfaces regenerated using glycine·HCl pH 1.5.
- High stability: XanTec’s Protein AG sensor chips help minimize assay costs and maximize reusability.
- Oriented immobilization: High ligand activity is maintained via directed immobilization through the Fc region.
| Product code2 | PAGP | PAGD200L | PAGHC30M | PAGHC200M |
|---|---|---|---|---|
| Base coating | 2D, ultra-short bioinert CM-dextran (high density) | 3D, 200 nm bioinert CM-dextran (low density) |
3D, 30 nm bioinert polycarboxylate (medium density) |
3D, 200 nm bioinert polycarboxylate (medium density) |
| Capture immobilization capacity [µRIU]3 | ≈ 5,000 | ≈ 12,000 | ≈ 6,000 | ≈ 11,000 |
| Ligands | Fc region of all human IgG subclasses, strong affinity for mouse IgG2a, IgG2b, and IgG3, and total IgG from cow, goat, sheep, and rabbit | |||
| Analytes |
|
|
|
|
| Intended purpose |
|
|
|
|
1 All illustrations are schematic representations and are not drawn to scale; dimensions, densities, and spatial relationships do not reflect actual physical or chemical proportions.
2 Table includes a selection from XanTec’s full Protein AG sensor chip portfolio.
3 Based on specific binding of 100 µg/mL IgG in phosphate-buffered saline (PBS), with 1 µRIU corresponding approximately to 1 RU.