DBCO–modified sensor chips
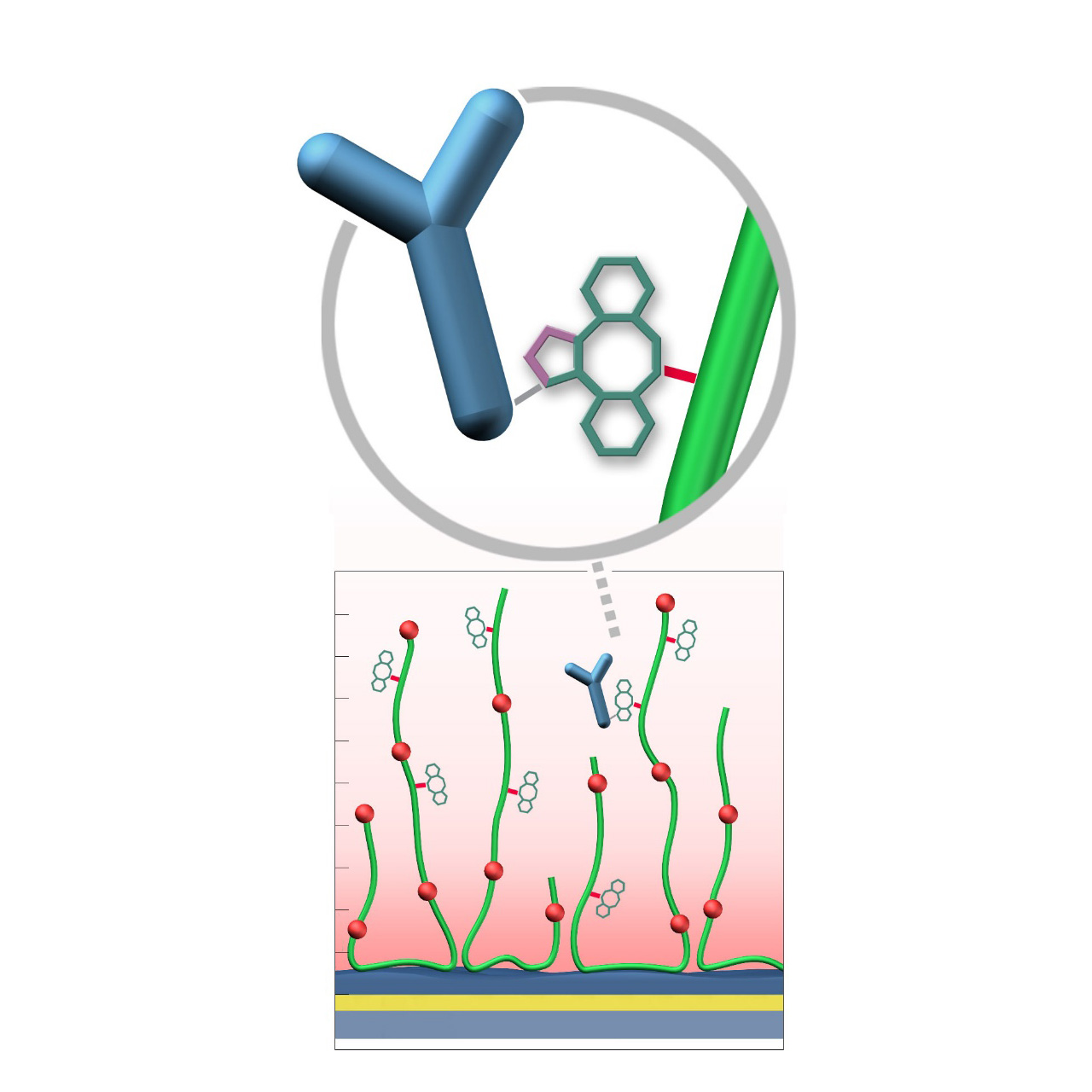
XanTec’s DBCO-modified sensor chips (DC) are coated with a dibenzocyclooctyne (DBCO)-derivatized bioinert carboxymethyl-dextran (CMD) matrix grafted onto a hydrophilic adhesion promoter on a gold support. Ligands containing a compatible Click partner—such as an azide group—can be covalently attached rapidly and selectively via strain-promoted azide-alkyne cycloaddition (SPAAC, Click reaction).
Unlike classical EDC/NHS amine-coupling, DBCO/azide Click chemistry is not time-critical; both reaction partners remain stable for weeks across pH 4–10. Their high selectivity minimizes side reactions and eliminates the need for quenching steps1. Consequently, Click-based immobilization is reliable, convenient, and operationally flexible. Due to the fast reaction kinetics, coupling under physiological conditions is feasible; however, maximum immobilization densities may be somewhat lower. Therefore, electrostatic preconcentration remains advantageous for achieving high ligand loading.
DBCO-modified surfaces exhibit a higher hydrophobicity than azide-modified counterparts or unmodified CMD coatings. This can lead to increased nonspecific binding, which is at least partially mitigated by the presence of nonionic detergents (e.g., Tween) in the running buffer. DBCO-modified sensor chips are most useful when the ligand naturally contains azide functionalities. Otherwise, the azide-modified sensor coating combined with a DBCO-modified ligand is generally preferred due to its higher intrinsic bioinertness.
Key features:
- Versatile ligand coupling: Efficient and selective Click reaction enables covalent attachment of azide-functionalized ligands. Unlike classic amine coupling, ligand density can be iteratively increased after initial coupling, allowing fast optimization of immobilization levels and efficient sensor chip use.
- Superior efficiency: High efficiency of the Click reaction permits ligand immobilization even under physiological conditions.
- Azide functionalization: Ligands can be conveniently azide-modified using labeling strategies analogous to biotinylation; site-specific conjugation via azide-containing unnatural amino acids is also feasible.
- Application versatility: Suitable for kinetic, equilibrium, and concentration analyses, as well as diverse screening applications in drug discovery.
| Product code | DCD50L | DCD200M |
|---|---|---|
| Base coating | 3D, 30 nm bioinert CM-dextran (low density) | 3D, 200 nm bioinert polycarboxylate (medium density) |
| Covalent immobilization capacity [µRIU]3 | ≈ 3,000 | ≈ 11,000 |
| Recommended ligands |
|
|
| Recommended analytes |
|
|
| Intended purpose |
|
|
1 This is valid only when the analyte lacks its own Click-reactive functionality; otherwise, unintended covalent attachment may occur.
2 All illustrations are schematic representations and are not drawn to scale; dimensions, densities, and spatial relationships do not reflect actual physical or chemical proportions.
3 Covalent immobilization capacity was assessed by injecting 100 µg/mL azide-modified bovine serum albumin (BSA) in 5 mM sodium acetate buffer (pH 5.0), with 1 µRIU corresponding approximately to 1 RU.