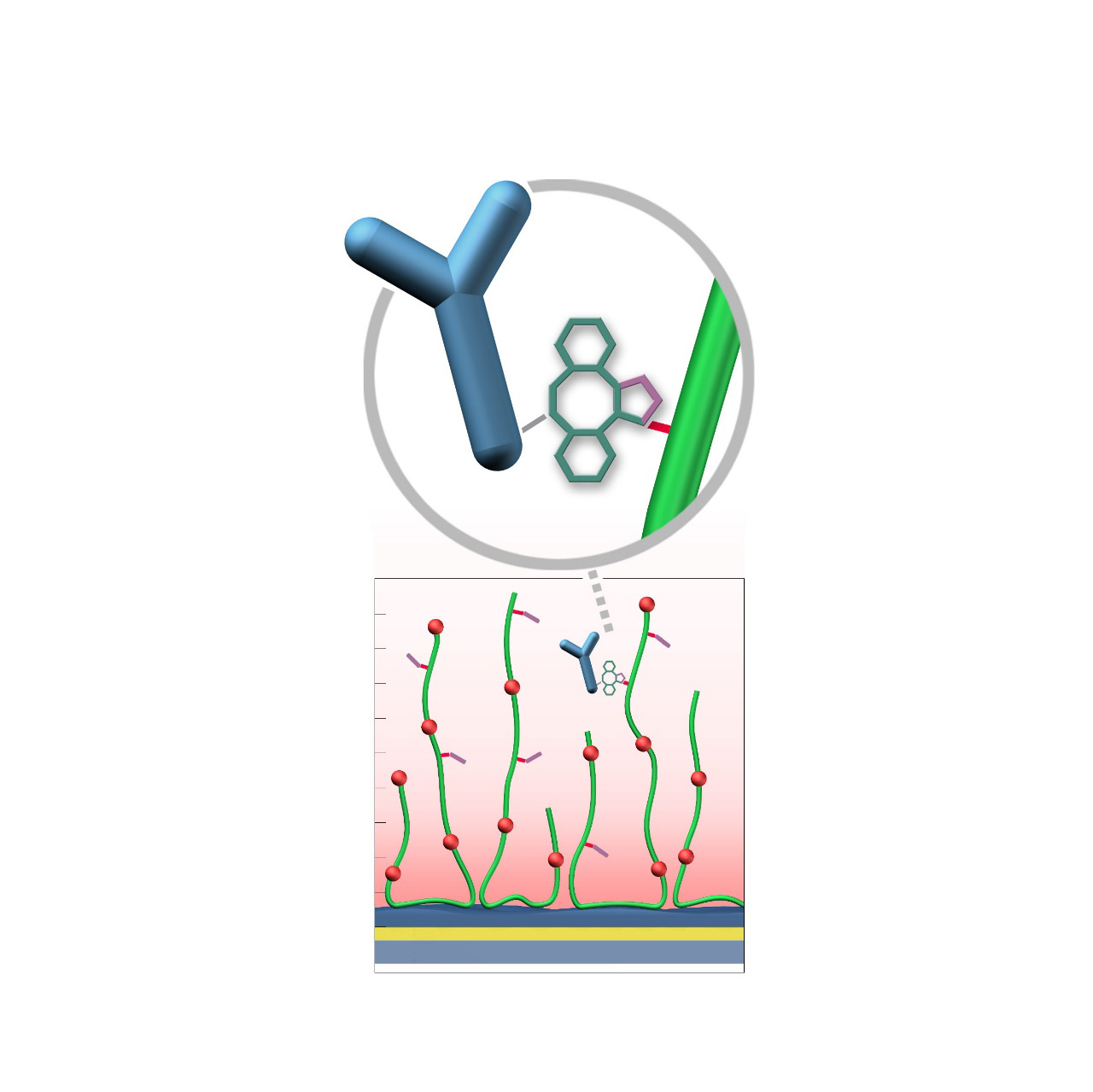
Azide–modified sensor chips
XanTec’s AZ sensor chips are coated with a bioinert polycarboxylate matrix (CMD or HC), pre-functionalized with low-molecular-weight azido groups grafted onto a hydrophilic adhesion promoter on a gold support. Ligands containing a compatible Click partner—such as dibenzocyclooctyne (DBCO)—can be covalently attached in a fast and selective manner via strain-promoted azide-alkyne cycloaddition (Click reaction).
Unlike classical EDC/NHS chemistry, DBCO/azide coupling is not time-critical; both reaction partners remain stable for days to weeks at pH 4–10. Their high selectivity minimizes side reactions and eliminates the need for quenching steps1. Consequently, immobilization via Click coupling is reliable, convenient, and flexible. Owing to rapid reaction kinetics, coupling under physiological conditions is feasible, although maximum immobilization densities may be somewhat lower; thus, electrostatic preconcentration remains advantageous for achieving high ligand loading.
The AZ sensor chip portfolio spans electrostatic immobilization capacities from approximately 5,000 µRIU (AZP) to 40,000 µRIU (AZHC1500M), covering analytes from large viruses to small organic fragments. The surface-bound azido groups are charge-neutral and highly hydrophilic, further enhancing the overall bioinertness of the coating.
Key features:
- Versatile ligand coupling: The bioorthogonal Click reaction enables clean, efficient covalent attachment of DBCO-functionalized ligands without EDC/NHS activation, hydrolysis, or quenching steps. Ligand density can be iteratively increased after initial coupling, allowing fast optimization of immobilization levels. Coupling under physiological conditions is also feasible.
- DBCO functionalization: Ligands can be conveniently DBCO-modified using standard labeling protocols similar to biotinylation. Oriented immobilization via site-specific DBCO conjugation is also supported.
- Wide immobilization range: From several thousand to ~40,000 µRIU, suitable for analytes from whole cells and viruses to fragments < 300 Da.
- Bioinert nanoarchitecture: Combining a hydrophilic adhesion promoter, hydrated polycarboxylate matrix (CMD or HC), and charge-neutral, hydrophilic surface azido groups minimizes nonspecific binding.
- Application versatility: Suitable for kinetic, equilibrium, and concentration analyses, as well as diverse screening applications in drug discovery.
| Product code3 | AZP | AZD200M | AZHC200M |
|---|---|---|---|
| Base coating | 2D, ultra-short bioinert CM-dextran (high density) | 3D, 200 nm bioinert CM-dextran (medium density) | 3D, 200 nm bioinert polycarboxylate (medium density) |
| Covalent immobilization capacity [µRIU]4 | ≈ 2,000 (5,000) | ≈ 13,000 (30,000) | ≈ 13,000 (26,000) |
| Recommended ligands |
|
||
| Recommended analytes |
|
|
|
| Intended purpose |
|
|
|
1 This is valid only when the analyte lacks its own Click-reactive functionality; otherwise, unintended covalent attachment may occur.
2 All illustrations are schematic representations and are not drawn to scale; dimensions, densities, and spatial relationships do not reflect actual physical or chemical proportions.
3 Table includes a selection from XanTec’s full AZ sensor chip portfolio.
4 Covalent immobilization capacity was assessed by injecting 100 µg/mL DBCO-modified streptavidin in 5 mM sodium acetate buffer (pH 5.0), with 1 µRIU corresponding approximately to 1 RU. Electrostatic preconcentration capacity is shown in brackets.